Share this:
Organic chemistry 1 Questions
1. Use the flow chart below to answer the questions that follow:
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
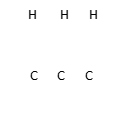
![]()
(a) What observation would be made in process K?
(b) Name another conditions necessary for process J to take place
(c) Give the name of substance V
2. But-z-ene undergoes hydrogenation according to the equation given below
CH3CH = CHCH3
(g) + H2(g) CH3CH2CH2CH3(g)
(a) Name the product formed when but-z-ene reacts with hydrogen gas
(b) State one industrial use of hydrogenation
3. Write the structures of the following compounds:-
(a) But—2-yne
(b) 2,2-dimethylpropane
4. a)What is meant by Isomerism?
b) Draw and name two Isomers of butane.
5. Study the information in the table below and answer the questions that follow:
Ion | No. of protons | No. of electrons |
P3- Q+ R2+ | 7 19 12 | 10 18 10 |
a) Write the electron arrangement of element P.
b) Give the group and period to which elements Q and R respectively.
Q ……………………………………………………
R ……………………………………………………
6. Compound W reacted with chlorine to form compound X only. The structural formula of
X is shown below:
CH3 – CH – CH – CH3
Cl Cl
(a) Give the structural formula and name of compound W
(b) Name compound X ……………………………………………………………………
7. In petrol chemical industries, long chain alkanes are broken down in to simpler substances
in a process called cracking
a) Why is cracking necessary?
b) State the two conditions required in cracking
c) Draw the structure of 1-chloro-2, 2-dimethylpropane
8. In a reaction an alcohol K was converted to hex-1-ene
a) Name reagent and condition necessary for the reaction in 6 (a) above to occur
9. (a) Give the IUPAC systematic names of compounds Q and R
Q: CH2CHClCHlCH2CH3
R: CH3CHClCH2ClCH3
(b) The organic compounds Q and R in (b) above, are formed when one mole of hydrocarbon
N reacts with two moles of hydrogen chloride gas;
(i) Structural formula of N
(ii) The IUPAC systematic name of N
10. Distinguish between the isotopes and isomers
11. Polymerisation of ethene takes place as shown in the equation below
Name the type of polymerisation undergone by ethene in the reaction above
12. (a) State Gay Lussac’s law
13. 10cm3 of methane (CH4) gas is exploded with 150cm3 of air containing 20% oxygen
and 80% nitrogen. The products were allowed to cool to room temperature. What will
be the total volume of the gases at the end of the reaction?
14. Give the open structures of:-
(i) 3-chlorohex-l-yne
(ii) CH3OH
15. A fixed mass of gas occupies 105cm3 at -14ºC and 650mmHg pressure. At what temperature in
degrees Celsius will it have a volume of 15cm3 if the pressure is adjusted to 690mmHg pressure?
16. Write an equation for the reaction that takes place between ethene and concentrated
Sulphuric (VI) acid
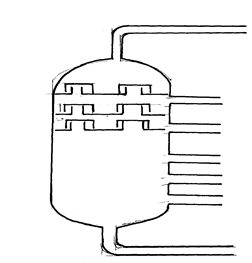
17. Petroleum (crude oil) is a mixture of several compounds which are separated in a Changamwe refinery by means of apparatus as shown below:
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
(a) (i) What is the name of the apparatus above
(ii) What is the name of the process which is used in separation of crude oil
(iii) What physical property of compounds in the mixture does the separation depend
(iv) Use the letter A to G to describe where the following could be formed:.
I. The fraction that represents gases
II. The fraction that represents the largest molecules
III. The fraction that represents liquids with the lowest boiling points
(b) State the use of product produce at
G………………………………………………………………………………………
C……………………………………………………………………………………….
(c) Draw apparatus for the separation of the product produce at D and water
18. Study the flow chart below and answer the questions that follow:-
(i) Give the name of the substance CH CH …………………………………………
(ii) To which group of hydrocarbons does the substance in (i) above belong?
(iii) Give two reagents that can be used to prepare the substance named in (i) above
(iv) State two physical properties of the substances in (i) above
(v) Give the names to the process in step I and 2
(vi) Write an equation to show how substance A is formed
(iv) Identify substance B ……………………………………………………
19. The diagram below represents a large-scale fractional distillation plant used to separate
the components A, B, C and D in a mixture
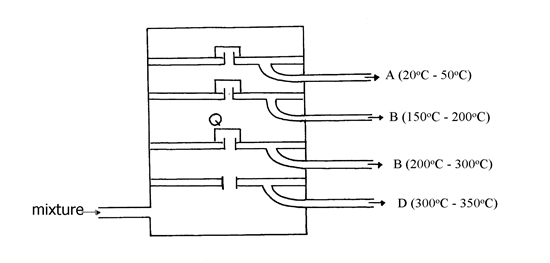
(a) The components have the following average relative molecular masses not necessarily in that
order; 282, 184, 44 and 128.
(a) (i) What is the physical state of B at the position marked Q?
(ii) Which component has an average relative molecular mass of 128? Explain
(iii) State with a reason whether C is pure or impure
(iv) Explain how the mixture is separated into its components
(v) Name two naturally occurring mixtures that are separated using this process
20. The diagram below represents a large-scale fractional distillation plant used to separate
the components A, B, C and D in a mixture
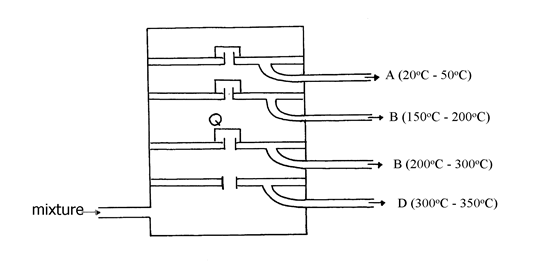
(a) The components have the following average relative molecular masses not necessarily in that
order; 282, 184, 44 and 128.
(a) (i) What is the physical state of B at the position marked Q?
(ii) Which component has an average relative molecular mass of 128? Explain
(iii) State with a reason whether C is pure or impure
(iv) Explain how the mixture is separated into its components
(v) Name two naturally occurring mixtures that are separated using this process
21. a) The table below gives information about the major constituents of crude oil. Study it and
answer the questions that follow:
Constituent | Boiling point oC |
Gases Petrol Kerosene Diesel Lubricating oil Bitumen | Below 40 40-175 175-250 250-350 350-400 Above 400 |
i) Which of the constituents of crude has molecules with the highest number of carbon
atoms? Explain
ii) Name the process you would use to separate a mixture of petrol and diesel and explain how
the separation takes place
iii) Explain why the constituents of crude oil do not have a sharp boiling point
iv) Name the gas that is likely to be a constituent of crude oil and write its formula
b) i) What condition could cause a poisonous gas to be formed when kerosene is burnt.
Explain
ii) Give one use of bitumen
22. (a) The set-up below was used to prepare ethyne gas
(i) Identify solid E
(ii) Complete the diagram to show how the gas can be collected
(iii) Write an equation to show how the gas is formed
(iv) Complete the equation below: )
C2H2 + 2I2
(v) What is the role of sand in the experiment?
(b) (i) Explain the meaning of esterification
(ii) Complete the equation below :
CH3COOCH3 + H2O
(iii) What type of reaction is occurring above
(c) Given the reaction:
![]()
C8H18 N + C2H4
(i) Identify substance:
F………………………………… N………………………………
(ii) Name the process represented above?
(d) Give one use of substance N
23.
(i) Name another source of hydrogen apart from electrolysis of water
(ii) What conditions are necessary for step III to occur?
(iii) Write the equation for the formation of colourless gas Q
(iv) Give one use of nitric (V) acid
(b) State and explain the observations that would be made if a sample of copper metal is
heated with concentrated nitric (V) acid
24. (a) Give the systematic names of the following compounds:-
(i) CH2 = C – CH3 ………………………………………………………………..
Br
(ii) CH3CH2CH2C CH ………………………………………………………..
(b) State the observations made when buton-l-ol reacts with:-
(i) Acidified potassium dichromate (VI) solution
(ii) Potassium metal
(c) Ethanol obtained from glucose can be converted to ethene as shown below:-
C6H12O6 C2H5OH C H2 = CH2
Name and describe the processes that take place in steps I and II
(d) Compounds A and B have the same molecular formula C3H6O2. Compound A librates
Carbon (IV) Oxide on addition of aqueous sodium carbonate while compound B does not.
Compound B has a sweet smell. Draw the possible structures of:-
(e) Give two ways how the disposal of polymers such as polychloroethene by burning pollutes
the environment
25. (a) Name the following compounds (CH3)3 C CH2 CH2 CH3
Use the flow chart below to answer the questions that follow:-
(b) (i) Name the following :-
I. Gas S ……………………………………………………………….……. ( )
II. Gas P …………………………………………………………………
III. J ……………………………………………………………………….
(ii) Name the processes involved in the following steps:
I. Step I …………………………………………………………………………..
II. Step II …………………………………………………………………………….
III. Step III …………………………………………………………………………….
(iii) Write a chemical equation for the complete combustion of substance M
(iv) Name the condition and reagent in step III
Condition ……………………………………………………………………………………
Reagent …………………………………………………………………………………….
(v) Calculate the mass of salt R that would be formed by using 21.9 tonnes of N when it reacts
with excess sodium hydroxide ( C= 12.0 H= 1.0 Na = 23)
(vi) Draw the structure of polymer K
II. State one use of the above polymer
………………………………………………………………………………………………….
![]() (c) (i) Name the class to which the following cleansing agents belong:-
(c) (i) Name the class to which the following cleansing agents belong:-
i) ……………………………………………………
(ii) ……………………………………………………….
II. Which cleaning agent above is not environmental friendly? Explain
26. The molecular formula of a hydrocarbon is C6H14. The hydrocarbon can be converted into two
other hydrocarbon as shown by the equation below:
C6H14 C2H6 + X
(i) Name and draw the possible structural formula of X
(ii) State and explain the observations that would be made if a few drops of bromine water
were added to a sample of X
(iii) Write an equation for the complete combustion of C3H8
27. (a) Give the names of the following
(i) CH3CH2CH3
(ii) CH3CCCH3
(b) Ethene is used in making polyethene bag in a process called polymerization
(i) Name the type of polymer that is formed when ethane polymerise
(ii) Describe a simple chemical test that can be used to identify ethane gas in the laboratory
(c) Study the information in the table below and answer the questions that follow:-
No. of carbon atoms | R.M.M of the Hydrocarbon |
2 3 4 | 28 42 56 |
i. Write the general formula of the hydrocarbons in the table above
ii. Determine the molecular of a hydrocarbon with 5 carbon atoms and draw its structural formula
Molecular formula
Structural formula
(d) Study the scheme below and answer the questions that follow

(i) Name the reagents in
Step I ……………………………
Step II ……………………………
Step IV ………………
(ii) Write an equation for the complete combustion of CH CH
(iii) Give two uses of CH4
28. Give the systematic names of the following compounds;
i)CH3 = C-CH3
CH3
ii)CH3CH2CH2C≡ CH
29. Study the data given in the following table and answer the questions that follow. The letters
are not the actual symbols of elements.
Element | Number of protons | Melting point | Bpt oC |
A | 11 | 98 | 890 |
B | 12 | 650 | 1110 |
C | 13 | 60 | 2470 |
D | 14 | 1410 | 2360 |
E | 15 | 442 590 | 280 |
F | 16 | 113 119 | 445 |
G | 17 | -101 | -35 |
H | 18 | -189 | -186 |
(i) State and explain the trend in melting point in A B C
(ii) Explain why the melting point and boiling points of element D is the highest
(iii) Explain why the element represented by letter E has two melting point values
(iv) Write down the chemical formula between element C and sulphate ions
(v) Name the chemical family in which H belong and state one use of the element
(vi) What is the nature of the oxide of the elements represented by letters C and F?
30. a) The table below gives information about the major constituents of crude oil. Study it and
answer the questions that follow:
Constituent | Boiling point oC |
Gases Petrol Kerosene Diesel Lubricating oil Bitumen | Below 40 40-175 175-250 250-350 350-400 Above 400 |
i) Which of the constituents of crude has molecules with the highest number of carbon
atoms? Explain
ii) Name the process you would use to separate a mixture of petrol and diesel and explain how
the separation takes place
iii) Explain why the constituents of crude oil do not have a sharp boiling point
iv) Name the gas that is likely to be a constituent of crude oil and write its formula
b) i) What condition could cause a poisonous gas to be formed when kerosene is burnt.
Explain
ii) Give one use of bitumen
31. Study the information in the table below and answer the questions that follow
Number of carbon atoms per molecule | Relative molecular mass of the hydrocarbon |
2 3 4 | 28 42 56 |
i) Write the general formula of the hydrocarbons in the table
ii) Predict the relative atomic mass of the hydrocarbons with 5 carbon atoms
iii) Determine the relative atomic mass of the hydrocarbon in (ii) above and draw its
structural formula (H=1.0, C=12.0)
32. Substance “M” with a general formula C2Hy burnt in chlorine gas with a red flame producing
a cloud of black specks and colourless gas G.
(a) State the collective name for compounds which ‘M’ belongs
(b) With reason, state the identity of the black specks and colour gas “G”.
33. 2.63g of a solution of sodium chloride at 20.0oC was reacted with silver nitrate. After filtration,
washing and drying, 2.36g of silver chloride was obtained. Determine the solubility of sodium
chloride at 20.0oC . (Na=23, Cl= 35.5, Ag = 108)
(b) Determine the number of moles of carbon (IV) Oxide gas produced when sodium
carbonate reacted with dilute sulphuric (VI) acid (Molar gas volume =24dm3)
34. Write down all the isomers of but-z-ene and give their IUPAC names
35. (a) A hydrocarbon compound Z decolourizes bromine liquid in the presence of light but
does not decolourize acidified potassium manganate (VII). Name and draw the structural
formula of the eighth member of this homologous series
36. (a) What is meant by isomerism?
(b) Draw and name two isomers of Butyne
Organic chemistry 1 Answers
1. a) Bromine decolorized immediately in ethane gas √1
b) Temperature between150°C – 250°C or temperature of 180°C
c) Carbon (IV) oxide or CO2(g) √
2. (a) Butane
(b) Manufactures of cooking fats and margarine
3. (a)
(b)
![]()
4. a) Existence of cpds with the same molecular formula but different structural
formula/arrangement of atoms
b)
n – butane/ ½ 2 – butane/½
l – butane/ But-2-ene 2 – methyl
But-1-ene prop-1-ene
5. a) 2.5
b) Q Group 1 ½, Period 4 ½
R Group 2 ½, Period 3 ½
6. a) H H H H
W- H C – C = C C- H
H H
7. a) To produce simpler hydrocarbons of industrial importance e.g. ethane which is widely used
b) Elevated temperature / high temperature 900 C
Catalyst
c) HC – C CH3
8. a) Reagent concentrated sulphuric acid
Condition temperature 180o C
9. a) H2 CHCL CHCLCH2CH3
Name: 2, 3 dichloropentane
b) i) Structural Formula
H H H
H – C = C – C = C – C – H
H H H
ii) IUPAC name
pent – 1,3 – diene
10. Isotopes are atoms of the same element with same atomic number but different mass numbers
while isomers are compounds with the same molecular formula but different structural formula
11. Addition polymerization. √1
![]() 12. (a) When gases combine they do so in volume which bear a simple ratio to one another and to
12. (a) When gases combine they do so in volume which bear a simple ratio to one another and to
the product if gaseous under standard temperature and pressure
13. CH4 + 2O2(g) CO2(g) + 2H2(l)
![]() 10cm3 20cm3 10cm3
10cm3 20cm3 10cm3
Volume of O2 = 20 x 150
![]() 100
100
= 30cm3
Remaining volume of O2 = 30-20=10cm3
![]() Total volume of the gases = 20+10+10
Total volume of the gases = 20+10+10
= 40cm3
H H H H
![]()
14. (i) H – C C – C – C – C – C – H
Cl H H H
H
![]()
(ii) H – C – O – H
![]() H
H
15.
T2 = 690 X 15 X 259√
650 X 105
= 39.3K√
= – 233.7° C√
16. CH2 =CH2g + H2SO4(L) ________ CH3CH2OSO3H(aq)
√ 1 mark
1 7 (a) i) Fractional Column.
ii) fractional distillation.
iii)different boiling points.
IV I A II F III B
b) G – road making or water proofing
C jet fuel or cooking and lighting.
![]() 18. (i) ethyne
18. (i) ethyne
(ii) Alkynes – because it has triple bond between the two carbon atoms
(iii) Water is calcium carbide
![]() (iv) – Colourless, odourless
(iv) – Colourless, odourless
![]() -less denser than air
-less denser than air
– Insoluble in water but soluble in organic solvents
(v) Hydrogenation
(vi) Halogenations
(vii)
![]()
(viii) Carbon (IV) Oxide
(ix) Nitrogen I Oxide (N2O)
![]()
![]() 19. (a) (i) Gas /vapour
19. (a) (i) Gas /vapour
![]() (ii) B – It has the second lowest boiling point thus second lowest molecular mass
(ii) B – It has the second lowest boiling point thus second lowest molecular mass
![]()
![]() (iii) C is impure since it boils over a range of temperature
(iii) C is impure since it boils over a range of temperature
![]() (iv) It is boiled heated and the vapour of the components condense at different temperatures
(iv) It is boiled heated and the vapour of the components condense at different temperatures
![]() (v) – Liquid air
(v) – Liquid air
– Crude oil
![]()
![]() 20. (a) (i) Gas /vapour
20. (a) (i) Gas /vapour
![]() (ii) B – It has the second lowest boiling point thus second lowest molecular mass
(ii) B – It has the second lowest boiling point thus second lowest molecular mass
![]()
![]() (iii) C is impure since it boils over a range of temperature
(iii) C is impure since it boils over a range of temperature
![]() (iv) It is boiled heated and the vapour of the components condense at different temperatures
(iv) It is boiled heated and the vapour of the components condense at different temperatures
![]() (v) – Liquid air
(v) – Liquid air
– Crude oil
21. a) i) Bitumen it has the highest boiling point
ii) Fractional distillation; during distillation petrol would distill off at 175C, while diesel
will distill at 350C
iii) Each component is a mixture of hydrocarbons which have different boiling points
iv) Methane, CH4, EthaneC2H6 propane, C3H8, Butane C4H10
b) i) Burning in limited amount of air will produce carbon monoxide (carbon (II) Oxide)
which is poisonous
ii) Manufacture of Tar used in road tarmacking sealing of leakages on roofs
22. A. (i) Calcium carbide – CaC2
(ii) Over water method
(iii) CaC2(s) + 2H2O(s) + 2H2O(l) Ca(OH)2(aq) + C2H2(g)
(iv) C2H2 + 2I2 C2H2I2
(v) The reaction if highly exothermic hence sand helps to absorb excess heat.
B. (i) A reaction in which an organism acid reacts with an alkanol to form a sweet smelling
compound called ester.
(ii) CH3COOCH3 + H2O CH3COOH + CH3OH
(iii) Hydrolysis
C (i) F – Aluminium oxide – Al2O3
N – C6H14 – Hexane
(ii) Cracking
D. A fuel
23. i) Cracking of crude oil fractions. √1
ii) Temp – 400 – 5000c
Pressure – 200 – 500 atmospheric Any 2 = 1
Catalyst – Finally divided iron.
iii) 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(l)
iv) – Manufacture of nitrate fertilizers. √1
– Manufacture of explosives.
– Purification of metals.
b) – Red brown gas
√1 with pungent irritating smell due to reduction of HNO3 to NO2
– Blue √1 solution due to formation of Cu (NO3)2
24. (a) (i) 2-bromo propene or 2- bromo prop-i-ene
(ii) Pent-i-ene
(b) (i) Changes form orange to Green
(ii) Effervescence//bubbles of gas produced
(c) Step 1
– Fermentation of glucose
Glucose broken down in obscene of oxygen using enzymes
- Dehydration of ethanol; using concentrated sulphuric (VI) acid and high temperature
of 170oC
Step II
- Dehydration of ethanol; using concentrated sulphuric (VI) acid and high temperature of 170oC
(d) Compound A
(e) – release chlorine gas which destroy ozone layer
– Chlorine gas combines with vapour in atmosphere to form acid rain which destroy vegetation
– Chlorine gas can cause respiratory diseases
25. (a) (i) 2,2 – dimethyl pentane
(b) I carbon IV oxide.
II Hydrogen gas.
III Propane.
- I Hydrogenation.
II Neutralization
III substitution
(iii) CH3CH2CH2Ol + 902 (g) 6 CO2
(g) + 8 H2O(l)
(iv) Condition Presence of U.u light
Reagents – Chlorine gas
(v) CH3CH2 CooH + NaoH CH3 CH2 COONa + H2O(c)
Mole ratio :
74 tones of acid 96 tones of salt
21.9 21.9 X 96 = 28.4 tones
74
Or 21.9 = 0.29 moles of salt
74
= 0.29 X 96 = 28.4 tones
(iv) I H CH
C C
H H n
(ii) use in making – Plastic crates plastic boxes plastic ropes
( c) I (i) soap detergent
(ii) Soap less detergent
II Soap less Detergent – non biodegradable.
![]() 26. (i) But-i-ene Or (accept any 1)
26. (i) But-i-ene Or (accept any 1)
But-z-ene
![]()
![]() (ii) Bromine water is decolourised because X is unsaturated or has a (-C = C-) double bond.
(ii) Bromine water is decolourised because X is unsaturated or has a (-C = C-) double bond.
(iii) C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(l)
27. a) i) Propane
ii) But- 2 –yne
b) i) Ploythene
ii) Bubble pass ethane gas in acidified KMnO4 or acidified K2Cr2OT
c) i) CnH2n
ii) @5H10
d) i) Step I – hydrogen
Step II – Hydrogen chloride
Step IV – Sodalime
ii) 2CH = CH(g) + O2(g) ________ 4CO2(g) + 2H2O(L)
- A fuel
- Manufacture of methanol
- Manufacture of methanol
28. i) 2 – Methylprop – l ene 1 mark
ii) Pent – L – yne 1 mark [Total 12 marks]
- The melting point increases from A to C this is due to increase in number delocalized electron
hence increase in the strength of metallic bond.
D forms a giant structure with strong covalent bonds. Hence high melting.
It exhibits allstrophy ie may exist as two different form in the same state.
C2 (so4)3
Noble gases or inert
Used in filament bubls
Used to produce an inert atmosphere in high temperature inetallurgical processes e.g welding.
C is amphoteric oxide
F acidic it is non –metal oxide.
Ethene
H
H
C= C
H
H
Acidified potassium Manganate VI abromine water it from a colourless solution
CH2CH2 + H2 CH3CH3
Nickel catalyst
30. a) i) Bitumen it has the highest boiling point
ii) Fractional distillation; during distillation petrol would distill off at 175C, while diesel
will distill at 350C
iii) Each component is a mixture of hydrocarbons which have different boiling points
iv) Methane, CH4, EthaneC2H6 propane, C3H8, Butane C4H10
b) i) Burning in limited amount of air will produce carbon monoxide (carbon (II) Oxide)
which is poisonous
ii) Manufacture of Tar used in road tarmacking sealing of leakages on roofs
31. i) CnH2n, where n = No. of carbon atoms
ii) 70
iii) CsH10, CH3CH=CHCH2CH3
OR CH3CH2CHCH2= CH2
32. (a) Hydrocarbon. √1
(b) Black specks is carbon
Colourless gas is steam √1 3
Hydrocarbon burn in air to form carbon √ ½ and water√½
33. NaCl(aq) AgNO3(aq) NaNO3(aq) + AgCl(s)
Moles of AgCl= Mass
R.F.M
= 2.36
![]() 143.5
143.5
= 0.016446moles
Mole ratio Nacl: AgCl
![]() 1 :1
1 :1
Moles of NCl = 0.61446moles
Mass of NaCl = RFM x moles
= 58.5 x 0.016446
![]() = 0.962091g
= 0.962091g
Mass of solvent (water) = 2.63 – 0.962091
![]() = 1.667909g
= 1.667909g
1.667909g of water dissolves 0.962091g of NaCl
100g of water dissolves = 100 x 0.962091g
![]() 1.667909
1.667909
= 57.68/100g of water
33. 24000cm3 = 1mol
![]() 80cm3 = 80×1
80cm3 = 80×1
![]() 2400
2400
= 0.00333moles
34.. (i) CH3CH = CHCH3 – But-z-ene
(ii) CH3C = CH2 2– methyl 1 prop-I-ene
CH3
(iii) CH2 = CHCH2CH3 – But-I-ene
35. (a) Octane
![]() or CH3CH2CH2CH2CH2CH2CH2CH3
or CH3CH2CH2CH2CH2CH2CH2CH3
36. a) Existence of same molecular formula but different structural formula √1
b) i)