Share this:
6. Structure of the atom and the periodic table Questions
1. In an experiment an unknown mass of anhydrous sodium carbonate was dissolved in water and
the solution made up to 250cm3. 25cm3 of this solution neutralized 20cm3 of 0.25M nitric acid.
(Na = 23.0 C = 12.0 O = 16.0)
Calculate:
(a) Moles of Nitric acid used
(b) Moles of sodium carbonate in 25cm
of the solution
(c) Mass of unknown sodium carbonate used
2. Element A has atomic mass 23 and element B has atomic mass 7 and also have 12neutorns and
4 neutrons respectively.
(a) Write the electronic arrangement of A and B
(b) Which element has higher ionization energy? Explain
3. The table below shows the relative atomic masses and the percentage abundance of isotope
M1 and M2 of element M.
Relative atomic mass | % abundance | |
M1 | 62.93 | 69.09 |
M2 | 64.93 | 30.91 |
Calculate the relative atomic mass of element M
4. (a) Element V has two isotopes. Two thirds of V and one third of V . What is the
relative atomic mass of element V?
(b) The following refers to element Y
Isotope | A | B | C |
Isotope mass | 54 | 56 | 57 |
Given that isotope C contains 31 neutrons in its nucleus find the number of protons in isotope B
5. The table below shows the relative atomic masses and the percentage abundance of the isotopes
L1 and L2 of element L.
Relative atomic mass | % abundance | |
L1 L2 | 62.93 64.93 | 69.09 30.91 |
Calculate the relative atomic mass of element K.
6. An element M has two isotopes M and M . The relative atomic mass of the
naturally occurring is 63.55. Calculate the percentage of each isotope
7. An oxide of element G has the formula as G2O3
(a) State the valency of element G
(b) In which group f the periodic table is element G?
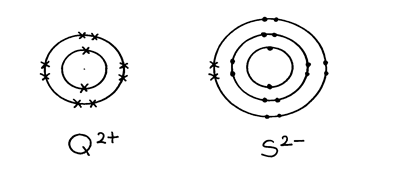
8. The table below gives information about the ions T+ and Z2-
Ion | T+ | Z2- |
Electron arrangement | 2.8 | 2.8.8 |
Number of neutrons | 12 | 16 |
(a) How many protons are there in the nucleus of ?
(i) Element T?
(ii) Element Z?
(b) Determine the relative formula mass of the compound formed between T and Z
(c) State two conditions under which the compound would conduct electricity
9. Carbon and silicon belong to the same group of the periodic table, yet Carbon (IV) oxide
is a gas while silicon (IV) oxide is a solid with a high melting point. Explain this difference
10. An ion of oxygen is larger than oxygen atom. Explain
11. Copper (II) oxide and charcoal are black solids. How would you distinguish between the
two solids?
12. (a) Element X is found in period III and group IV. It consists of two isotopes 28X and QX.
A sample of X was found to consist of 90% of 28X.If the relative atomic mass of X is 28.3,
work out the number of neutrons in QX
(b) Draw an electrochemical cell for the above cell
13. Study the table below and answer the questions that follows:- (Letters are not the actual
symbols of element)
Element | Electronic arrangement | Electrical conductivity |
L1 | 2.8.2 | Higher electrical conductivity |
L2 | 2.8.1 | High electrical conductivity |
L3 | 2.8.3 | Highest electrical conductivity |
L3 has the highest electrical conductivity. Explain
14. Define the term melting point of a substance
15. Use the information in the table below to answer the questions that follow.
(The letters do not represent the actual symbols of the elements).
(a) Which two letters represent the same element? Give a reason
(b) Give the number of neutrons in an atom of element R
16. The table below gives some elements in the periodic table. Use it to answer the questions that
Follow. The letters do not represent the actual symbols of the elements.
Element | A | B | C | D | E |
Atomic number | 12 | 13 | 14 | 15 | 16 |
Which of the above letters represent:
a) A metallic element which forms ions with the smallest ionic radius? Explain
b) A non metallic element with the largest bbatomic size? Explain
17. The grid below is part of the periodic table. Use it to answer the questions that follow:
(The letters are not the actual symbols).
A B
C D G E
F
a) Write down the formula of the compound formed between C and A.
b) Which element has the same electron arrangement as the stable ion of:
(i) F ……………………………. (ii) A ……………………..
c) Element Q has atomic number 15. Indicate its position on the grid.
d) Explain how the atomic radii of the following compare:
(i) C and F
(ii) C and D
e) Write the type of bond present in a compound formed between D and A.
f) Compound C and G were completely burned in oxygen.
(i) Write down equations to show the combustion of each of the elements.
(ii) State whether each of the oxides (i) above is basic or acidic.
18. The following flow chart shows the industrial manufacture of Nitric (V) acid.
a) Identify substance B, C, E and F.
b) Describe what happens in the catalytic chamber.
c) State what takes place in chamber D.
d) 60 – 65% nitric (V) acid is produced in the absorption chamber. Describe how the acid can be
concentrated.
e) State why nitric (V) acid is stored in dark bottles.
f) Copper reacts with nitric (V) acid and not hydrochloric acid. Explain.
19. The number of protons, neutrons and electrons in atoms A to F are given in the table below
the letters do not represent the actual symbol of the elements:-
Atoms | Protons | Neutrons | Electrons |
A B C D E F | 3 9 12 17 17 18 | 4 10 12 18 20 22 | 2 10 12 17 17 18 |
(a) Choose from the table the letters that represent:
(i) An atom of a metal …………………………………………………………………
(ii) A neutral atom of a non-metal ……………………………………………….
(iii) An atom of a noble gas …………………………………………………..
(iv) A pair of isotopes …………………………………………………………………….
(v) A cation …………………………………………………………………….
(b) The grid below shows a part of the periodic table. The letters do not represent the actual
symbols.
Use it to answer the questions that follow:-
(a) How do the atomic radius of element X and Y compare
(b) (i) Using crosses (X) to represent electrons, draw the atomic structure of element Q
(ii) State the period and the group to which element Q belong
(c) (i) The ionic configuration of element G is 2.8 G forms an ion of the type G-1.
Indicate on the grid, the position of element G.
(ii) To which chemical family does element G belong?
(iii) State one use of element U
(iv) What is the nature of the compound formed between K and U
20. (a) Study the table below and answer the questions that follow.
Particle | Atomic number | Ionic configuration | Formula of oxide | Atomic radii | Ionic radii |
P | 4 | ………….. | …………… | 0.110 | 0.031 |
Q | …………… | 2.8.8 | QO | 0.200 | 0.099 |
R | …………… | 2.8.8 | R2O | 0.230 | 0.133 |
S | 17 | 2.8.8 | S2O7 | 0.099 | 0.181 |
T | 16 | …………… | ……… | 0.104 | 0.231 |
(i) Complete the table above
(ii) From the table, choose the most reactive metal. Explain
(iii) Which element is the most electronegative. Explain
(iv) Using dots (.) and crosses (x) to represent electrons, show the bonding in the chloride of Q
(v) Explain the solubility of element T in water
(b) (i) Why is aluminium used to make utensils yet it is a reactive metal?
(ii) Distinguish between valency and oxidation number
21. a) Work out the oxidation number of phosphorous in the following compound H3PO3
b) Study the equation below:
Mg(s) + 2H2O(l) Mg(OH)2(aq) + H2 (g)
Which species has undergone oxidation .Explain
22. The grid below represents part of the periodic table. The letters do not represent the actual
symbols of the elements. Study it and answer the questions that follow:
L |
| L | ||||||
M | P | T | J | U | X | |||
N | Q | R | S | V | Y | |||
W | ||||||||
(a) Explain why element L appears in two different groups in the grid above
(b) State the name of the chemical family to which P and Q belong
(c) Write the formula of the compound formed between P and V
(d) Compare the melting points of Q and S. Explain
(e) Identify an element whose oxide dissolves in both acids and alkalis
(f) Write the equation for the burning of T in excess air
(g) Using dots (•) and cross (x) to represent electrons, draw a diagram to illustrate bonding
in the sulphide of Q
(h) State one use of element X
23. The grid below represents part of the periodic table. Study it and answer the questions that follow:
(a) (i) Identify the element that gains electrons most readily
(ii) Which of the metal is most reactive? Explain
(iii) What name is given to the family of elements to which elements X and T belong?
(iv) Explain why:-
(I) Ionic radius of Q is larger than that of M
(II) Atomic radius of Q is greater than that of S
(v) Which of the element in the table does not have the ability to form an ionic or covalent
bond? Explain
(vi) Give the formula of the compound formed between R and Z
24. The grid below is part of the periodic table. The elements are not represented by their actual symbols. Use the information to answer the questions that follow.
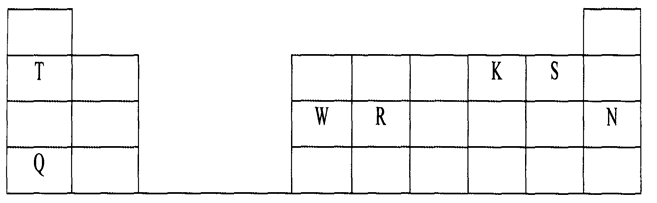
a) (i) Which is the most reactive
(I) Non — metal? *
Explain
(II) Metal?
Explain
(ii) Name the family to which elements T and Q belongs.
(iii) Write the formula of the compound formed when W reacts with S.
(iv) Name the type of bond and structure formed when elements R and K react.
(v) Explain why element N doesn’t form compounds with other elements.
(vi) Compare the atomic radii of T and Q. Explain.
25. Study the data given in the following table and answer the questions that follow. The letters
are not the actual symbols of elements.
Element | Number of protons | Melting point | Bpt oC |
A | 11 | 98 | 890 |
B | 12 | 650 | 1110 |
C | 13 | 60 | 2470 |
D | 14 | 1410 | 2360 |
E | 15 | 442 590 | 280 |
F | 16 | 113 119 | 445 |
G | 17 | -101 | -35 |
H | 18 | -189 | -186 |
(i) State and explain the trend in melting point in A B C
(ii) Explain why the melting point and boiling points of element D is the highest
(iii) Explain why the element represented by letter E has two melting point values
(iv) Write down the chemical formula between element C and sulphate ions
(v) Name the chemical family in which H belong and state one use of the element
(vi) What is the nature of the oxide of the elements represented by letters C and F?
26. An element W has an atomic number 13.
a) Write the electronic configuration of the most stable ion of W
b) Write the formula of the oxide of the element W
27. Identify the particles that facilitate the electric conductivity of the following substances
(i) Sodium metal
(ii) Sodium Chloride solution
(iii) Molten Lead Bromide
28. Compare with a reason the atomic radius of Sodium to that of Aluminum.
29. Study the information in the table below and answer the questions that follow:
Ion | No. of protons | No. of electrons |
P3- Q+ R2+ | 7 19 12 | 10 18 10 |
a) Write the electron arrangement of element P.
b) Give the group and period to which elements Q and R respectively.
Q ……………………………………………………
R ……………………………………………………
30. Ethanol is a liquid at room temperature but does not conduct electricity. Explain.
31. Electronic configuration for elements represented by P, Q, R and S are:-
P= 2.8.6, Q= 2.8.2, R= 2.8.1 D= 2.8.8.
- Select the element which forms
(i) A double charged ion
(ii) A soluble carbonate
32. The table below gives information on four elements by letters K, L, M and N. Study it
and answer the questions that follow. The letters do not represent the actual symbol of
the elements.
Element | Electron arrangement | Atomic radius (nm) | Ionic radius (nm) |
K L M N | 2.8.2 2.8.7 2.8.8.1 2.8.8.2 | 0.136 0.099 0.203 0.174 | 0.065 0.181 0.133 0.099 |
(a) Which two elements have similar properties? Explain
(b) What is the most likely formula of the oxide
of L?
(c) Which element is non-metal? Explain
33. Study the information given below and answer the questions that follow:
Element | Atomic radius (nm) | Ionic radius (nm) | Formula of oxide | Melting point of oxide (oC) |
A | 0.364 | 0.421 | A2O | -119 |
D | 0.830 | 0.711 | DO2 | 837 |
E | 0.592 | 0.485 | E2O3 | 1466 |
G | 0.381 | 0.446 | G2O5 | 242 |
J | 0.762 | 0.676 | JO | 1054 |
(i) Write the formula of the compound formed when J combined with G
(b) Explain why the melting point of the oxide of E is higher than that of the oxide of G
6. Structure of the atom and the periodic table Answers
1. Na2CO3 + 2HNO3 ____ 2NaNO3 (L) + CO(q) + H2O(C)
Mole ration 1 : 2
a) Moles of HNO3 in 20cm3 = 20/1000 x 0.25
= 0.005 moles
b) Moles of Na2CO3 in 25cm3 = ½ of 0.005 moles
= 0.0025
c) If 25cm3 = 0.0025 moles
in 250cm3 = ?
250 x 0.0025
25
= 0.025 moles
RFM of Na2CO3 = 106
I mole of Na2CO3 = 106g
0.025 moles = ?
0.025 x 106
1
= 2.65g of Na2CO3
2. (a) A= 2.8.1
![]() B= 2.1
B= 2.1
(b) B
Strong attraction of the outermost energy level electron to the nucleus make it difficult
to remove This is due to smaller atomic radius compared to A
Or – Outermost electrons are closer to the nucleus hence higher force of attraction
3. R.A.M = (62.93 x 69.09) + (64.93 x 3091)
100
= 4347.834 + 2006.99
100
= 63.5482
63.5
![]() 4. (a) R.A.M = (33 x 2) + (30 x 1)
4. (a) R.A.M = (33 x 2) + (30 x 1)
![]() 3
3
99 = 33
3
(b) Number of electrons of C = 57-31 = 26
Number of electrons of B is the same as for C = No. of Protons
![]() B = 26 protons
B = 26 protons
5. 69.09 x 62.93 + 30.91 x 64.93 1
100 100
43.4783 + 20.0698 1
= 63.548 ≃ 63.55 1
6. 63 x + 65 (100 – x) = 63.55
100
63x + 6500 – 65x = 6355
2x = 6355 – 6500
2x = -145
X = 72.5
% abundance of 63 M = 72.5%
65 M = 27.5%
7. a) Valency of G is 3
b) G is a group 3 element
8. a) i) 11 protons
ii) 16 protons
b) Formula of compound = T2Z
Mass number of T = 11+ 12 = 23
Mass number of 2 = 16+16 = 32
Formula Mass of T2Z = ( 23×2) + 32 = 78
c) – When molten
– When in aqueous solution
9. Silicon (iv) Oxide has giant atomic structure with strong covalent bond holding the atom
together. These require a lot of energy to break, hence it has high melting point. Carbon (IV) Oxide has simple molecular structure with weakVan Der Waals forces holding the molecules together which require little energy to break, hence sublimes at low temperature and is a gas at room temperature and pressure
10. O2 2.8 O 2.6
The oxide ions has 2 extra electrons that causes greater electron repulsion than in oxygen atom
11. To separate samples of CUO and charcoal in test tubes, dilute mineral acid is added with
shakingCuO black dissolves to form blue solution ½
Charcoal does not dissolve in dilute mineral acids
12. (90 x 8) + 10Q = 28.3 (½mk)
100
100 x 2520 + 10Q = 28.3 x 100
100
2520 + 10Q = 2830 (½mk)
10Q = 2830 – 2520
10Q = 310
Q = 31
Electron arrangement of X = 284 (½mk)
Atomic No. = 14 ( ½mk)
No. neutrons = 31 – 14 = 17 (½mk)
13. L3 has delocalised electrons while the others has less
14. (a) Is a constant temperature at which a solid changed to a liquid/ A point at which a solid
changes to a liquid which a solid changes to a liquid without change in temperature.
15. (a) P √ ½ and S √ ½
√
They have the same atomic numbers. √ Both must be there to score 3
(b) 4 (7, -3) √
16. a) B√ ½ – its ion has a stronger nuclear charge than that of A√ 1
b) D√ ½ – has the weakest nuclear charge as compared to the other non- metals √ 1
17. (a) CA 1
(b) (i) E 1
(ii) B 1
(c) Period 3, ½ Group 2, ½
(d) (i) The atomic radius of F is greater than that of C1 because F has more energy levels.
(ii) The atomic radius D is smaller than that of C 1 because of increased positive charge
in the nucleus which attracts the electrons more. 1
(e) (i) Electrovalent bond ½
(ii) Covalent bond ½
(f) (i) 4C + O2 2C2O 1
G + O2 GO2
1
(ii) C2O is basic while 1
GO2 is acidic. 1
18. (a) B – ammonia gas 1
C – nitrogen (II) oxide (NO) 1
E – water 1
F – unreacted gases 1
(b) The mixture of ammonia and air is passed through heated/ catalyst where ammonia (II) is
oxidized to nitrogen (II) oxide. 1
(c) Gases are cooled and air passed through heated/ catalyst where ammonia is further
oxidized to nitrogen(IV) oxide. 1
(d) Fractional distillation, ½
Water with a lower boiling point ½ than nitric (V) acid, distills left leaving the
concentrates acid.
![]() 19. (a) (i) C
19. (a) (i) C
![]() (ii) D or E
(ii) D or E
(iii) F
![]() (iv) D or E
(iv) D or E
(v) A
![]()
![]() (vi) D
(vi) D
![]() (b) Atomic radius of Y is smaller than that of X. The effective nuclear charger in Y is greater
(b) Atomic radius of Y is smaller than that of X. The effective nuclear charger in Y is greater
than in X hence outer electrons strongly pulled to the centre reducing the radius.
![]()
![]() (b) (i)
(b) (i)
![]() (ii) Period – 3 Group – IV
(ii) Period – 3 Group – IV
(c) (i) On the grid (period 2 Group 7)
![]()
![]() (ii) Halogen
(ii) Halogen
(iii) – Used in hospitals with patients with breathing difficulties
![]() – Used by mountain climbers and deep sea divers
– Used by mountain climbers and deep sea divers
(iv) Basic
20. A (i) P – ionic configuration – 2
– Formula of oxide – PO
Q – Atomic number – 20
R- Atomic number – 19
T – Ionic configuration – 2.8.8
Formula of oxide – TO2
(ii) R – Has the largest atom with one outer electron hence easily loses it.
(iii) S – is the smallest atom of a non-metal with a deficit of only one electron hence
easily gains.
(iv)
![]()
![]()

![]()
![]()
![]()
(v) T is insoluble – It has a molecular structure/non-metal
(B)(i) It is coated with an un reactive layer of aluminium oxide which prevents it form reacting.
(ii) Valency – The number of electrons an atom gains or loses during a reaction.
Oxidation number – The resultant charge of an atom has after gaining or loosing electrons.
21. a) +3 + P = (-2×3)= 0
+3+P – 6 = 0
P = +3√
b) Mg- its oxidation state increases from Zero to +2 √ 1 mark
22. a) Group 1 – Because √½ it has 1 electron in its outermost energy level.
Group 7 – It requires √½ 1 electron to fill its outermost energy level.
b) Alkaline earth metals √1
c) PV2
√1
d) Q has higher√½ m.p than J. Q has a giant metallic structure and strong metallic bonds. √½
While J has molecular structure and Vander
Waals forces which are easy to break. √½
e) R. √1
 f) T(s) + O2(g) TO2(g) √1
f) T(s) + O2(g) TO2(g) √1
g)
h) – Filling electric light bulb √1 accept any other correct one.
23. (a) (i) X Rj: If actual symbols are given.
(ii) Q. Rj. Actual symbols.
Explanation: It looses the outermost energy level most readily.
(iii) Halogens
(iv) I). Moving across a period there is increased nuclear charge.
II). Going down a group the energy levels increase in number.
(v) V- Explanation It has a complete outermost energy level/ Has a stable octet.
(vi) Z2R Rej. Interchange of letters, RZ2.
24. a) i) I S 1- It readily gain one electron on ionization1
II Q – It readily give out one electron on ionization1
ii) Alkali metals1
iii) WS31
iv) Bond – covalent ½
Structure – Giant atomic structure ½
v) It is stable. Cant remove nor add electrons on its outermost energy level
vi) T has a smaller radius than Q because it has fewer energy levels than Q
- The melting point increases from A to C this is due to increase in number delocalized electron
hence increase in the strength of metallic bond.
D forms a giant structure with strong covalent bonds. Hence high melting.
It exhibits allstrophy ie may exist as two different form in the same state.
C2 (SO4)3
Noble gases or inert
Used in filament bubls
Used to produce an inert atmosphere in high temperature inetallurgical processes e.g welding.
C is amphoteric oxide
F acidic it is non –metal oxide.
Ethene
H
H
C= C
H
H
Acidified potassium Manganate VI abromine water it from a colourless solution
CH2CH2 + H2 CH3CH3
Nickel catalyst
26. a) 2 : 8
b) W2O3
27. i) Delocalized electrons
ii) Mobile ions
iii) Mobile ions
28. – Sodium has a larger raius than aluminium
– Aluminium has more protons than sodium hence a more effective nuclear charge
than sodium
29. a) 2.5
b) Q Group 1 ½, Period 4 ½
R Group 2 ½, Period 3 ½
30. Ethanol contains molecules 1 which are not1 responsible for electrical conductivity. (2 mks)
31. a (i) Q
(ii) R
![]()
![]() 32. (a) K and N because they have the same number of electrons on their outermost energy level
32. (a) K and N because they have the same number of electrons on their outermost energy level
(b) L2O7
(c) L1 because it has 7 electrons on the outermost energy level or reacts by gaining electrons
or the ionic radius is larger than the atomic radius (½mk)![]()
33. a) Formula; J5G2 √1
b) E form ironic structures due to ionic bonding in its oxide. While G form molecular
structure due to covalent bonding in it oxide